This does not alter the authors' adherence to all the PLOS ONE policies on sharing data and materials, as detailed online in the guide for authors.Ī number of antibody and non-antibody protein scaffolds are in development, which include the domain antibodies, V HH domains from camelids, scFv, Fab, Abdurins, Affibodies, Adnectins, Centryns, and Darpins. owns certain patents and patents applications covering the Abdurins and the EphA2 binder, however, there are no products in development or marketed products to declare. Hagemeyer by Baker IDI, Matt Harris and Amos Hedt by Clarity Pharmaceuticals, Ltd. Christopher Ullman, Pascale Mathonet, Arkadiusz Oleksy and Agata Diamandakis are employed by Isogenica, Ltd., Licia Tomei, Anna Demartis, Chiara Nardi, Sonia Sambucini and Antonino Missineo by IRBM Science Park, Karen Alt and Christoph E. This study was funded and sponsored by Research Corporation Technologies, Inc., the employer of Kurt R. The specific roles of all authors are articulated in the ‘author contributions’ section.Ĭompeting interests: The authors have the following interests. provided support in the form of salaries for the authors but did not have any additional role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript. Research Corporation Technologies, Inc., Isogenica, Ltd., IRBM Science Park, Baker IDI, Clarity Pharmaceuticals and VTU Technology Ltd. KRG and the authors made the decision to publish this work. and the research activities directed by KRG. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are creditedĭata Availability: All relevant data are within the paper as text, figures and tables.įunding: This study was funded and sponsored by Research Corporation Technologies, Inc. Received: FebruAccepted: JPublished: August 27, 2015Ĭopyright: © 2015 Ullman et al. van Raaij, Centro Nacional de Biotecnologia-CSIC, SPAIN (2015) High Affinity Binders to EphA2 Isolated from Abdurin Scaffold Libraries Characterization, Binding and Tumor Targeting. These data demonstrate the ability to isolate high affinity binders from the engineered Abdurin scaffold, which retain a long serum half-life, and specifically target tumors in a xenograft model.Ĭitation: Ullman C, Mathonet P, Oleksy A, Diamandakis A, Tomei L, Demartis A, et al. The anti-EphA2 Abdurins localized in the tumors as early as 4 hours after injection and continued to accumulate up to 48 hours when the imaging was completed. 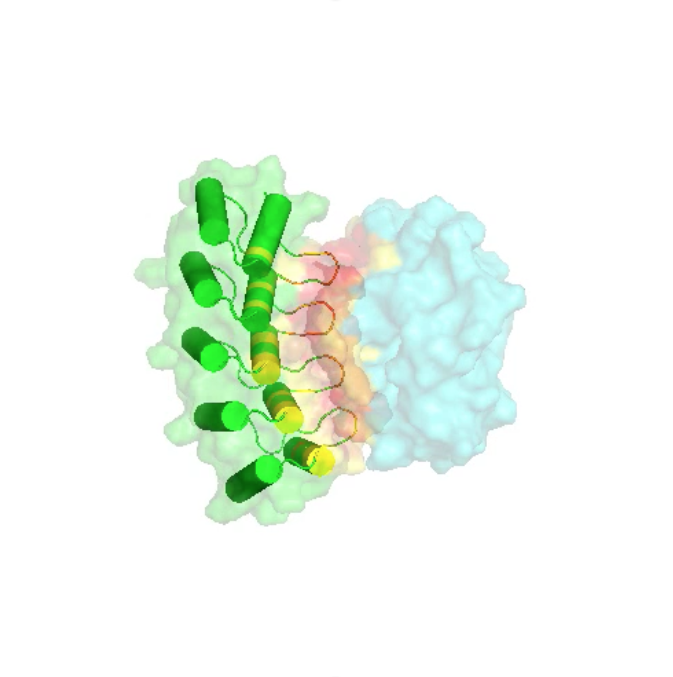 
Two different EphA2 binders were labeled with 64Cu, using a bifunctional MeCOSar chelator, and administered to mice bearing tumors from transplanted human prostate cancer cells, followed by PET/CT imaging. 
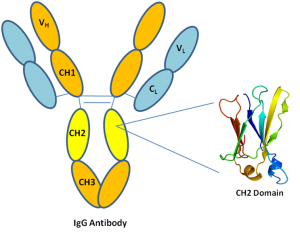
Further characterization confirmed binding of the Abdurins to both human and murine EphA2 proteins and exclusively to cell lines that expressed EphA2, followed by rapid internalization. Initial binders were reformatted into new maturation libraries and low nanomolar binders were isolated using cell-free DNA display, CIS display. Antigen binding regions were created by designing combinatorial libraries into the structural loops and Abdurins were selected using phage display methods. Here we present data on the construction of large, diverse, phage-display and cell-free DNA display libraries and the isolation of high affinity binders to the cancer target, membrane-bound ephrin receptor tyrosine kinase class A2 (EphA2). Previous studies established the prolonged serum half-life of Abdurins, the result of a retained FcRn binding motif. Abdurins are a novel antibody-like scaffold derived from the engineering of a single isolated CH2 domain of human IgG.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
